Images © Murray Robertson 1999-2011 Text © The Royal Society of Chemistry 1999-2011 The electrons become a little closer to the nucleus than expected and they also become slightly harder to remove than expected. As Einstein realised at such speeds strange things being to happen. 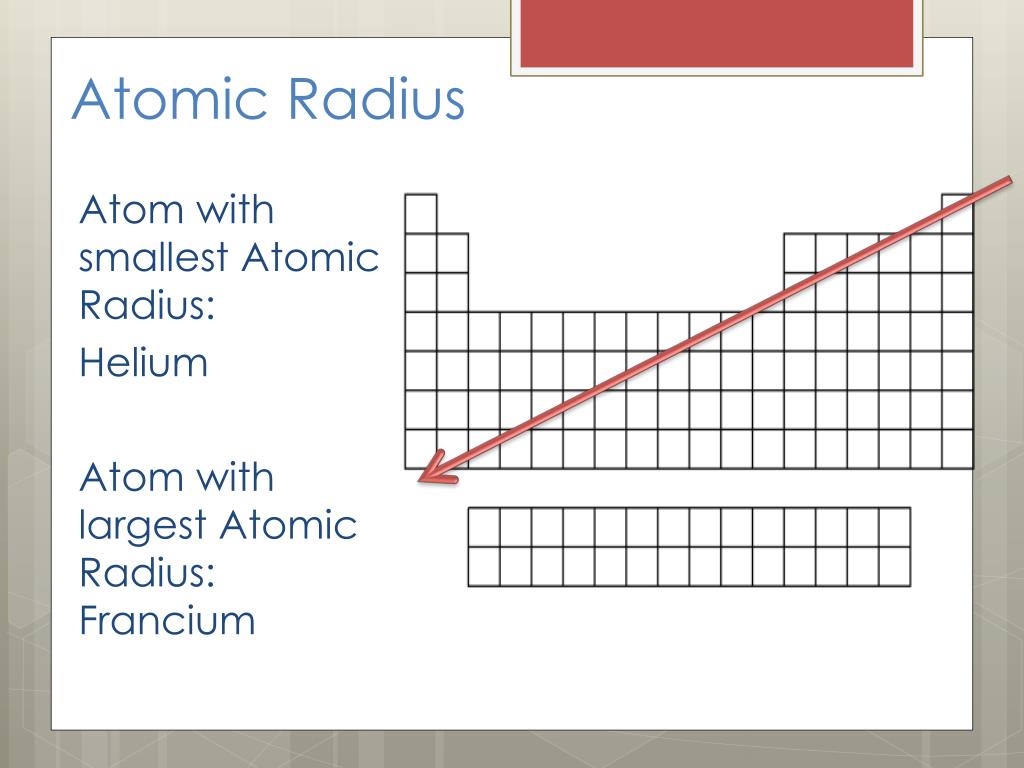
However, for the really heavy elements, the presence of so many positively charged protons in the nucleus has the affect of causing the electrons to move round at incredibly fast speeds approaching the sound of light. This is the trend for the elements lithium, sodium, potassium, rubidium and caesium. On descending a group in the periodic table, on average the outermost electrons get further and further away from the nucleus and as a result, become easier to remove from the atom. What's more, to the surprise of most chemists and going against the well-known trends of the periodic table, it turns out that francium is not the most reactive metal. It has been estimated that at any one time there is less than a kilogram of the element in the entire earth's crust. Whilst it is naturally occurring, or to be more precise, naturally formed - albeit briefly - during radioactive decay of other elements, the amount of francium on earth is tiny. Political stability of top reserve holderĪ percentile rank for the political stability of the country with the largest reserves, derived from World Bank governance indicators. The higher the value, the larger risk there is to supply.Ī percentile rank for the political stability of the top producing country, derived from World Bank governance indicators. The percentage of the world reserves located in the country with the largest reserves. The higher the value, the larger risk there is to supply. The percentage of an element produced in the top producing country. Low = substitution is possible with little or no economic and/or performance impact Medium = substitution is possible but there may be an economic and/or performance impact High = substitution not possible or very difficult. The availability of suitable substitutes for a given commodity. A higher recycling rate may reduce risk to supply. The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. The chemical properties of francium most resemble cesium.A vertical column in the periodic table. No weighable quantity of the element has been prepared or isolated. Because all known isotopes of francium are highly unstable, knowledge of the chemical properties of this element comes from radiochemical techniques. This is the only isotope of francium occurring in nature. The longest lived 223Fr (Ac, K), a daughter of 227Ac, has a half-life of 22 min. Thirty-three isotopes of francium are recognized. It has the highest equivalent weight of any element, and is the most unstable of the first 101 elements of the periodic system. While it occurs naturally in uranium minerals, there is probably less than an ounce of francium at any time in the total crust of the earth. 
It can also be made artificially by bombarding thorium with protons. Francium, the heaviest known member of the alkali metals series, occurs as a result of an alpha disintegration of actinium. Marguerite Perey of the Curie Institute, Paris. Back to Elements List Francium Atomic Number:ĭiscovered in 1939 by Mlle.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
